Image: Ideal Gas Law
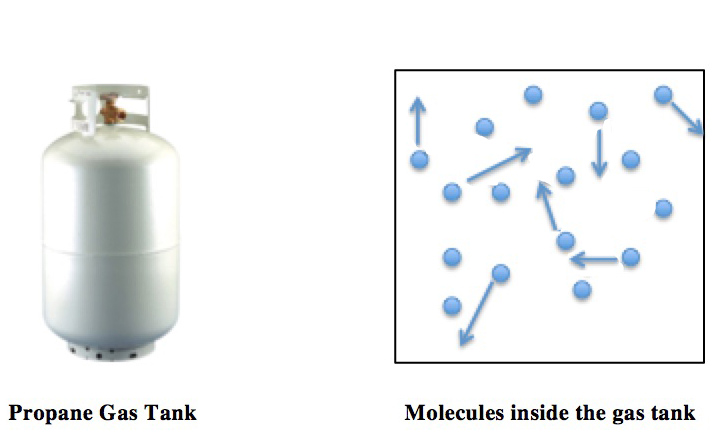
Description: Molecular collisions within a closed container (the propane tank) are shown (right). The arrows represent the random motions and collisions of these molecules. The pressure and temperature of the gas are directly proportional: as the temperature is increased, the pressure of the propane increases by the same factor. A simple consequence of this proportionality is that on a hot summer day, the propane tank pressure will be elevated, and thus propane tanks must be rated to withstand such increases in pressure.
Title: Ideal Gas Law
Credit: Own work
Author: BlyumJ
Usage Terms: Creative Commons Attribution-Share Alike 4.0
License: CC BY-SA 4.0
License Link: https://creativecommons.org/licenses/by-sa/4.0
Attribution Required?: Yes
Image usage
The following 2 pages link to this image: