Corrosion facts for kids
Corrosion is a natural process that slowly changes metals into a more stable form. Think of it like a metal trying to go back to how it was found in nature, often as a rock. This process gradually destroys materials, usually metals, through chemical reactions with their surroundings.
The most common type of corrosion happens when a metal reacts with something like oxygen or sulfur. A great example is rusting, which is when iron turns into iron oxides. You've probably seen rust on old bikes or metal fences. Many metal alloys, which are mixtures of metals, can corrode just by being exposed to moisture in the air.
Contents
What is Corrosion?
Corrosion is like a slow-motion attack on materials, especially metals. It's a chemical reaction where a metal loses tiny particles called electrons. This reaction often happens when the metal touches water and air (which has oxygen).
Imagine a shiny new metal object. Over time, if it's left outside, it might start to look dull or even get flaky. That's corrosion at work! It's a natural process, but it can cause a lot of damage to buildings, bridges, and even your bike.
How Does Rust Form?
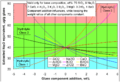
Rust is a common type of corrosion that happens to iron and its alloys, like steel. For rust to form, you need two main things: iron and oxygen. Water usually speeds up the process a lot.
When iron, oxygen, and water meet, a chemical reaction takes place. The iron atoms combine with oxygen atoms to create iron oxides, which we call rust. Rust is usually reddish-brown and crumbly, unlike the strong metal it came from.
Stopping Corrosion: How to Protect Metals
Since corrosion can damage important structures and objects, people have found many ways to protect metals. The field of Corrosion engineering focuses on understanding and preventing this process.
Removing Corrosion
Sometimes, if a metal has already started to corrode, you can clean it. For example, special chemicals like phosphoric acid (sometimes called "naval jelly") can be used to remove rust from tools or other metal surfaces. These chemicals react with the rust to break it down.
Making Metals Stronger Against Corrosion
Some metals naturally resist corrosion better than others. For instance, gold doesn't rust or corrode easily, which is why it stays shiny for a very long time.
There are also many ways to protect metals that are prone to corrosion:
- Painting: A layer of paint acts as a barrier, stopping oxygen and water from reaching the metal surface.
- Galvanizing: This process involves coating a metal, like steel, with a thin layer of zinc. Zinc corrodes instead of the steel, protecting it. This is called hot dip galvanizing.
- Sacrificial Anodes: For large structures like ships or pipelines, a piece of a more reactive metal (like zinc or magnesium) is attached. This "sacrificial" metal corrodes instead of the main structure, protecting it.
- Anodizing: This is a process that creates a thick, protective oxide layer on the surface of metals like aluminum. It makes the metal more durable and resistant to corrosion.
By using these methods, we can make sure that our bridges, cars, and even small tools last much longer and stay safe to use.
Images for kids
-
Corrosion on exposed metal, including a bolt and nut
-
Galvanized surface
-
Corroding steel gantry on an electrified railway line
-
Ozone cracking in natural rubber tubing
See also
 In Spanish: Corrosión para niños
In Spanish: Corrosión para niños