Aluminium iodide facts for kids
Template:Chembox LattConst Angle
Quick facts for kids Aluminium iodide |
|
|---|---|
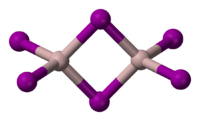 |
|
 |
|
|
Preferred IUPAC name
Aluminium iodide
|
|
| Other names | Aluminium(III) iodide Aluminum iodide |
| Identifiers | |
| CAS number | |
| PubChem | |
| EC number | 232-054-8 |
| SMILES | I[Al](I)I |
| Properties | |
| Molecular formula | |
| Molar mass | 0 g mol-1 |
| Appearance | white (anhydrous) or yellow powder (hexahydrate) |
| Density | 3.98 g/cm3 (anhydrous) 2.63 g/cm3 (hexahydrate) |
| Melting point | |
| Boiling point | |
| very soluble, partial hydrolysis | |
| Solubility in alcohol, ether | soluble (hexahydrate) |
| Structure | |
| Crystal structure | Monoclinic, mP16 |
| Space group | P21/c, No. 14 |
| Lattice constant | a = 1.1958 nm, b = 0.6128 nm, c = 1.8307 nm |
| Thermochemistry | |
| Std enthalpy of formation ΔfH |
-302.9 kJ/mol |
| Standard molar entropy S |
195.9 J/(mol·K) |
| Specific heat capacity, C | 98.7 J/(mol·K) |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) | |
Aluminium iodide is a special kind of chemical compound. It is made up of only two elements: aluminium and iodine. You might also hear it called Aluminium(III) iodide or Aluminium triiodide.
Contents
What is Aluminium Iodide?
Aluminium iodide is a substance that combines aluminium and iodine. It can exist in a few forms. The most common one is called anhydrous, which means it has no water. There's also a hexahydrate form, which means it has six water molecules attached.
What Does it Look Like?
The anhydrous form of aluminium iodide is usually a white powder. The hexahydrate form, which contains water, is typically a yellow powder.
How is it Made?
You can make aluminium iodide in a couple of ways. One way is to react aluminium metal directly with iodine. Another method involves using hydrogen iodide gas and letting it react with aluminium metal.
What are its Properties?
Aluminium iodide has some interesting properties:
- Formula: The anhydrous form is written as AlI3. The hexahydrate form is AlI3·6H2O.
- Weight: One molecule of anhydrous aluminium iodide weighs about 407.7 grams per mole. The hexahydrate form is heavier, at about 515.8 grams per mole.
- Density: The anhydrous form is quite dense, about 3.98 grams per cubic centimeter. The hexahydrate is less dense, around 2.63 grams per cubic centimeter.
- Melting Point: The anhydrous form melts at about 188 degrees Celsius. The hexahydrate form melts around 185 degrees Celsius, but it also starts to break down at that temperature.
- Boiling Point: The anhydrous form boils at about 382 degrees Celsius. It can also turn directly into a gas without becoming a liquid first, a process called sublimation.
- Solubility: Aluminium iodide is very good at dissolving in water. However, when it dissolves, it also undergoes a partial hydrolysis reaction. This means it reacts with water to form other substances. It can also dissolve in liquids like alcohol and ether.
See also
 In Spanish: Yoduro de aluminio para niños
In Spanish: Yoduro de aluminio para niños

