Rechargeable battery facts for kids
A rechargeable battery is a special kind of battery. You can use it many times because you can fill it up with electrical energy again. People also call them 'secondary cells'. Batteries you throw away after one use are called 'disposable batteries' or 'primary cells'. Just like other batteries, rechargeable ones can be recycled to help the planet.


Rechargeable batteries are great because you can use them over and over. This means less pollution since fewer batteries are thrown away. They can also save you money in the long run, as you buy them less often. However, they usually cost more to make. They also contain more toxic chemicals than disposable batteries.
You can find rechargeable batteries in many portable devices. These include MP3 players, mobile phones, and flashlights.
Contents
Where Are Rechargeable Batteries Used?


Rechargeable batteries power many different things. They are used in car starters and portable gadgets. They also power light vehicles like motorized wheelchairs, golf carts, and electric bicycles. Even large machines like forklifts use them.
These batteries are also important for backup power systems. They are used in large battery storage power stations. Newer types of rechargeable batteries are being developed for hybrid cars and electric cars. The goal is to make them cheaper, lighter, and smaller, while lasting longer.
Older rechargeable batteries used to lose their power quickly. They needed to be charged before you could use them for the first time. But some newer types, like low self-discharge NiMH batteries, can hold their charge for many months. They are often sold already charged to about 70%.
Large Battery storage power stations use rechargeable batteries to store electricity. They can store power when there is low demand. Then they release it during busy times when more power is needed. This also helps with renewable energy sources. For example, they can store solar power generated during the day to be used at night. Storing power helps reduce the amount of electricity a power plant needs to make at its busiest times. This saves money and reduces the need for extra power plants.
Small rechargeable batteries are found in many electronic devices. They power tools, appliances, and more. Bigger, heavy-duty batteries power electric vehicles. These range from scooters to trains and even ships. They are also used in systems that generate electricity locally.
How Rechargeable Batteries Charge and Discharge
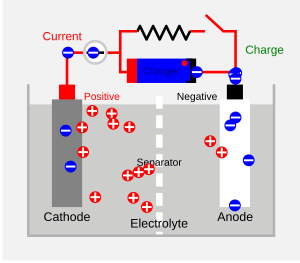
When you charge a rechargeable battery, a chemical process happens. Electrons move from one part of the battery to another. This creates an electric current that flows into the battery. The liquid or gel inside the battery, called the electrolyte, helps move tiny particles called ions between the two ends, called electrodes. This process stores electrical energy.
The energy to charge batteries usually comes from a battery charger. These often plug into a wall outlet (AC mains electricity). Some chargers can also use a car's 12-volt DC power outlet. The charger's voltage must be a bit higher than the battery's. This pushes the current into the battery. But it shouldn't be too high, or the battery could get damaged.
Charging a battery can take anywhere from a few minutes to several hours. Simple, slower chargers might take 14 hours or more to fully charge a battery. Faster chargers can charge batteries in two to five hours. The quickest ones can even do it in about fifteen minutes!
Fast chargers need smart features. They must be able to tell when a battery is full. They do this by checking the battery's voltage or temperature. This stops the charging process before the battery gets overcharged or too hot. The fastest chargers often have fans to keep the batteries cool. Battery packs designed for quick charging might even have a special temperature sensor. This sensor helps the charger protect the battery.
Different types of batteries need different ways of charging. For example, some batteries can be charged with a steady voltage. Others need a controlled current that slows down as the battery gets full. Charging a battery the wrong way can damage it. In serious cases, batteries can overheat, catch fire, or even burst.
Main Types of Rechargeable Batteries
Here are some common types of rechargeable batteries:
Related pages
Images for kids
See also
 In Spanish: Batería recargable para niños
In Spanish: Batería recargable para niños