Chemical burn facts for kids
A chemical burn happens when your skin or other body parts touch a strong chemical. These chemicals are often called corrosive substances. They can be strong acids or bases.
Chemical burns are like other burns. They can cause a lot of damage to your body's tissues.
Here are some things to know about chemical burns:
- They don't need heat to happen.
- They can happen right away when you touch the chemical.
- Sometimes, you might not notice the burn immediately.
- They can be very painful.
- The chemical might go deep into your body. It can damage parts under your skin, even if the skin surface looks okay at first.
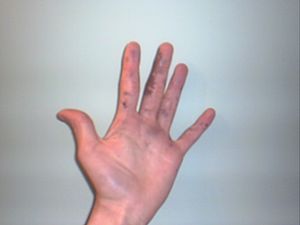
What do chemical burns look like?
What a chemical burn looks like depends on the chemical. You might feel itching or burning. Your skin might change color, becoming lighter or darker.
Sometimes, people have trouble breathing or cough up blood. Body tissue can also die, which is called tissue necrosis.
Common chemicals that cause burns include:
- sulfuric acid (H2SO4)
- hydrochloric acid (HCl)
- sodium hydroxide (NaOH)
- lime (CaO)
- silver nitrate (AgNO3)
- hydrogen peroxide (H2O2)
Each chemical can have different effects. For example, hydrogen peroxide can make a layer of skin look bleached. Nitric acid can turn skin yellow.
Chemical burns can happen in different ways:
- By touching the chemical on your skin or in your eyes.
- By breathing in chemical fumes.
- By swallowing the chemical.
Some chemicals, like hydrofluoric acid or sulfur mustard, can get into your body easily. They might not cause a burn right away. Instead, the burn and swelling can appear hours after you touched them.
People who work with chemicals, like in factories or labs, might get chemical burns. Hydrofluoric acid is very dangerous. It can get into your blood and react with important minerals like calcium. This can cause serious heart problems.
Other pages
See also
 In Spanish: Quemadura química para niños
In Spanish: Quemadura química para niños