Acorn banksia facts for kids
Quick facts for kids Acorn banksia |
|
|---|---|
 |
|
| Scientific classification | |
| Kingdom: | |
| (unranked): | |
| (unranked): | |
| Order: | |
| Family: | |
| Genus: | |
| Species: |
B. prionotes
|
| Binomial name | |
| Banksia prionotes |
|
| Synonyms | |
|
Sirmuellera prionotes (Lindl.) Kuntze |
|
Banksia prionotes, often called the acorn banksia or orange banksia, is a type of shrub or tree found in the Banksia family. It grows naturally in the southwest part of Western Australia. This plant can grow up to 10 meters (33 feet) tall, but it might be smaller in windy areas or further north.
The acorn banksia has dull green leaves with jagged edges. Its large, bright flower spikes start white and then open up to a bright orange color. People call it "acorn banksia" because its partly opened flowers look like an acorn. This tree is popular in gardens and is also important for the cut flower industry.
John Lindley, an English botanist, first described Banksia prionotes in 1840. It grows in sandy soils and is often the main plant in scrubland or low woodland. Birds help to pollinate its flowers. It provides food for many animals, including honeyeaters, especially in autumn and winter. In the Avon Wheatbelt area, it's super important for honeyeaters because it's the only plant flowering and making nectar at certain times of the year.
Contents
What Does it Look Like?
|
|
Banksia prionotes can grow as a tree up to 10 meters (33 feet) tall in the southern parts of its home. But in warmer, drier northern areas, it's usually a shorter tree or a spreading shrub, reaching about 4 meters (13 feet) high. It has thin, smooth or grooved bark that is mottled grey. Its young stems are covered in fine hairs. The dull green leaves are about 15 to 27 centimeters (6 to 11 inches) long and 1 to 2 centimeters (0.4 to 0.8 inches) wide. They have jagged, triangular edges and sometimes a wavy surface.
The flowers grow in a typical Banksia flower spike. This is a cluster of hundreds of small flowers packed tightly around a central stem. B. prionotes has cream-colored flowers with bright orange tips that only show when the flower fully opens. This opening process moves from the bottom to the top of the spike over several days. This makes the spike look like it's changing from cream to bright orange.
After flowering, the old flower parts fall off. Up to 60 seed pods, called follicles, can be seen on the stem. These pods are oval or oblong, about 14 to 20 millimeters (0.6 to 0.8 inches) long, and stick out a bit from the cone. Inside each pod are two seeds, separated by a woody divider. The seeds are dark, wedge-shaped, and have a thin, papery "wing."
The plant's root system has a main "sinker root" that goes straight down to the water table. This root can be up to 15 meters (49 feet) long if the water is that deep. It also has up to ten side roots that spread out horizontally, just below the surface. These side roots can grow more than 5 meters (16 feet) from the plant. In the wetter months, these side roots grow special "proteoid roots" that form dense mats near the surface. These help the plant get nutrients.
How is it Classified?
Banksia prionotes was first officially described by John Lindley in 1840. He likely based his description on plants collected by James Drummond in 1839. The name prionotes comes from Ancient Greek words meaning "saw" and "quality," referring to the saw-like, jagged edges of its leaves.
The most common names for B. prionotes are acorn banksia and orange banksia. People also call it saw-toothed banksia or golden banksia. The Noongar people, who are the traditional owners of the land, have a general name for Banksia plants in the Avon River area, which is Bwongka.
Scientists classify plants to show how they are related. Banksia prionotes belongs to the genus Banksia. Over the years, different botanists have placed it in various groups based on its features, like its flower spikes and leaf shapes. Today, it is considered part of a group of four closely related species that all have bright orange flowers. Its closest relative is Banksia hookeriana.
Scientists continue to study the relationships between Banksia species using DNA information. These studies generally agree that Banksia prionotes is closely related to Banksia hookeriana and other species in its group.
Plant Hybrids
Sometimes, different plant species can mix their genes and create a new plant called a hybrid.
Banksia prionotes and Banksia hookeriana
Banksia prionotes can easily create hybrids with Banksia hookeriana (Hooker's banksia) in controlled experiments. This shows their pollen is very compatible. A garden plant called B. 'Waite Orange' is thought to be one of these hybrids.

These hybrids have also been found in the wild, but only in places where the land has been disturbed, like along roadsides. The two parent species live in similar areas and are pollinated by the same birds. Normally, they don't hybridize much because they flower at different times of the year. B. prionotes usually finishes flowering by May, while B. hookeriana starts in June.
However, in disturbed areas, extra water and nutrients can make the plants grow bigger and flower for longer. This means their flowering times can overlap, allowing them to hybridize. These hybrid plants can produce seeds, just like their parents. This raises a question: could these hybrids eventually lead to the parent species losing their unique traits, especially if the hybrids are better at surviving in certain places?
Hybrids of Banksia prionotes and Banksia hookeriana have features that are in between the two parents. For example, they might be as tall as B. prionotes but branch out from the base like B. hookeriana. Their flowers and leaves are also a mix of both parents.
Other Possible Hybrids
A possible natural hybrid between B. prionotes and B. lindleyana (porcupine banksia) was found north of Kalbarri National Park. Hybrids of B. prionotes with B. menziesii (firewood banksia) have also been made by scientists and seen in nature.
Where it Lives
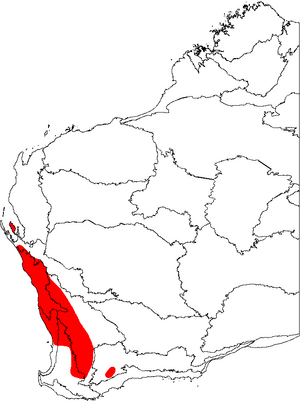
Banksia prionotes grows across a large part of the Southwest Botanical Province in Western Australia. It can be found along the west coast and further inland, from Shark Bay in the north to Kojonup and Jerramungup in the south.
It usually grows in tall shrubland or low woodland, mostly in the lower parts of sand dunes. It really likes deep white or yellow sand. It's very common in the kwongan heath of the Geraldton Sandplains north of Jurien, often being the most common plant there.
This plant is almost always found in the lower parts of sand dunes. One reason might be that it needs its long roots to reach the underground water. Another idea is that it can't survive in higher areas where fires happen too often. In fact, B. prionotes has started growing more along roadsides where fires are less common. However, it doesn't grow in areas that flood, because it doesn't like heavy soils and young plants die if flooded for too long.
How it Grows and Lives
Growth
The plant's root system, with its deep taproot and side roots, starts growing in the first year. The taproot grows straight down to the water table, which helps the plant get water during the dry summer. The side roots spread out horizontally and grow special "cluster roots" in the top layer of soil during autumn rains.
For the first eight years, the plant grows very quickly. After that, it slows down as it puts more energy into making flowers and seeds. Flowers start to appear in February and usually finish by the end of June.
How it Gets Food
Banksia prionotes has two special ways to get food and water. First, it's a phreatophyte. This means its long taproot reaches deep down to the water table, giving it a steady water supply during the dry summer months. This helps it survive and grow when other plants might struggle. The water from deep underground also provides the plant with important minerals.
Second, it has "cluster roots." These roots help it get enough nutrients from the poor soils where it grows. When autumn rains start, the side roots grow dense mats of these cluster roots in the top 20 centimeters (8 inches) of soil, where most minerals are found. These roots release chemicals that help dissolve minerals, making it much easier for the plant to take in nutrients like phosphorus, which is often hard to find in Australian soils.
These cluster roots take in water and minerals throughout winter and spring. But when the topsoil dries out in summer, they die off. The main side roots are protected from drying out by the water supplied by the deep taproot. This plant actually helps move water from deep underground up to the surface soil, a process called hydraulic redistribution. Making and then losing these cluster roots takes a lot of energy for the plant, but it's crucial for getting enough food in tough conditions.
How it Reproduces

Flowering starts in February and usually finishes by the end of June. Not many flowers bloom at once on each plant. Individual flowers open one by one, from the bottom to the top of the flower spike. More flowers open during the day, especially in the early morning when birds are most active.
Many birds that eat nectar, mainly honeyeaters, visit the flowers. These include the New Holland honeyeater and the red wattlebird. Lorikeets and insects like bees and ants also visit. However, only birds seem to be good at pollinating this plant. Insects don't seem to help with pollination, as flowers don't form seeds if birds are kept away.
Honeyeaters prefer to visit flowers that have just opened because they have the most nectar. As they drink, pollen sticks to their beaks and heads, which they then carry to other flowers. This is very effective: most pollen is gone from a flower within four hours of opening, and pollen is left on most of the flower's receptive parts.
Banksia flowers have a special structure where the end of the style (the female part) acts like a "pollen presenter." This might suggest that a flower could pollinate itself. However, B. prionotes seems to be mostly "self-incompatible." This means that even if pollen from the same plant lands on a flower, it usually can't fertilize it. So, even though birds often move between flowers on the same plant, leading to self-pollination, it doesn't often result in seeds.
The number of seed cones produced varies each year. While the flowering rate is low, the cones that do form usually have many seeds inside. Some of these seeds are eaten by insects, mainly a type of weevil.
Protecting the Plant
Banksia prionotes faces several threats. It is very sensitive to a plant disease called Phytophthora cinnamomi dieback, which kills plants. Wild populations are also harvested for the cut flower industry. Additionally, some of its habitat is being cleared for cities or farms.
Studies suggest that climate change could reduce its range by about 50% by 2080. Changes in fire regimes (how often and how intensely fires happen) have also affected it, likely leading to a decline in its numbers.
However, because Banksia prionotes is so widespread and common, it's not currently considered to be in danger as a whole species. Western Australia's Department of Environment and Conservation doesn't list it as rare.
Despite this, it's very important in the Avon Wheatbelt area. Here, it's a "keystone mutualist" because it's the only source of nectar for honeyeaters during a key part of the year. If B. prionotes disappeared from this region, the honeyeaters would also disappear. This would then affect many other plant species that rely on honeyeaters for pollination. The specific type of woodland where Banksia prionotes grows in the Avon Wheatbelt is considered a special ecological community that needs protection. It faces threats like land clearing, habitat loss, rising salty soils, grazing, weeds, changes in fire patterns, and the dieback disease.
Growing it at Home
Banksia prionotes is described as a "great ornamental species" because its bright, showy flower spikes make it a popular garden plant. It's excellent for attracting honeyeaters to your garden and sometimes flowers twice a year. There's a special dwarf (small) version available in Western Australia, called "Little Kalbarri Candles," which only grows to about 1 meter (3 feet) high.
It's fairly easy to grow in places with a Mediterranean climate (like parts of California or the Mediterranean region), but it doesn't do well in areas with high humidity in summer. It needs a sunny spot with soil that drains well and can handle moderate frost. You should prune it lightly, making sure not to cut below the green leaves, or it might become straggly as it gets older. Its seeds don't need any special treatment before sowing, and they usually sprout in 21 to 35 days.
This species is also great for producing cut flowers because its flowers grow at the end of long stems. It's one of the most popular banksias for this purpose, with commercial farms growing it in places like Israel, Victoria, South Australia, and Hawaii.
See also
 In Spanish: Banksia bellota para niños
In Spanish: Banksia bellota para niños