Charles Janet facts for kids
Quick facts for kids
Charles Janet
|
|
|---|---|

Janet around 1925
|
|
| Born |
Charles Janet
15 June 1849 Paris
|
| Died | 7 February 1932 (aged 82) Voisinlieu (Beauvais)
|
| Alma mater | École Centrale Paris |
| Known for | Left-step periodic table of chemical elements |
| Spouse(s) |
Berthe Marie Antonia Dupont
(m. 1877) |
| Children | 7 |
| Parents |
|
| Scientific career | |
| Fields | |
| Signature | |
Charles Janet (born June 15, 1849 – died February 7, 1932) was a brilliant French engineer, inventor, and scientist. He was interested in many different areas, from tiny insects to the building blocks of the universe – atoms! He is especially known for creating a unique version of the periodic table of chemical elements, which he called the "left-step periodic table."
Contents
Life and Work of Charles Janet
Charles Janet finished his engineering studies at the École Centrale Paris in 1872. For a few years, he worked as a chemist and engineer in factories in places like Puteaux and Rouen. In 1877, he married Berthe Marie Antonia Dupont and joined her family's company, which made bone buttons and fine brushes. He worked there for the rest of his life, but he always found time to explore his passion for science.
Janet was a very curious person. He studied many things, including the tiny heads of ants, wasps, and bees. His detailed drawings and photos of these insects were amazing! He also studied plants and wrote about how living things change over time, a process called evolution.
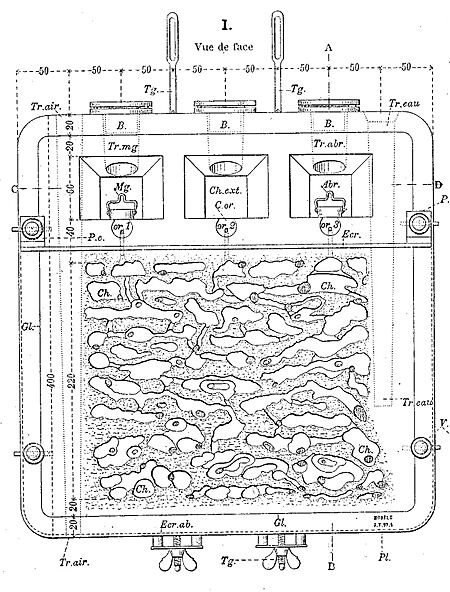
He was also an inventor. He designed many of his own tools, like the formicarium. This special ant farm uses two panes of glass to let you see an entire ant colony at work. Later in his life, around 1927, he became fascinated with the periodic table and developed his own unique way of organizing the chemical elements.
Charles Janet's Scientific Discoveries
While working at the factory, Charles Janet also went to university at the Sorbonne starting in 1886. He joined important science groups like the French Entomological Society, which studies insects, and the French Zoological Society, which studies animals. He was a top student and earned his doctorate in natural sciences in 1900, focusing his research on ants. Even before he finished his studies, the French Academy of Sciences recognized his work and gave him awards. In 1899, he became the president of the French Zoological Society.
In 1900, he showed off his improved artificial ant nests at a big exhibition in Paris. People were very interested in watching the ants, and journalists wrote about his inventions. In 1909, he received another award, the Cuvier Prize, for his important work in zoology.
Exploring Earth's Past: Geology and Paleontology
Charles Janet also loved exploring the Earth's history. He studied the Paris Basin region, especially its chalk layers, which are made of ancient sea creatures. He organized trips for university students to learn about geology around the city of Beauvais.
He collected an amazing number of fossils and ancient artifacts, about 50,000 items in total! Many of these fossils came from places that are now hard to reach or have disappeared. He even found a special way to protect the delicate shells from these ancient layers. His collection included many sea urchins (called echinoderms) and three new types of ancient squid-like creatures called belemnites in the chalk near Beauvais. He named these new species Actinocamax grossouvrei, Actinocamax toucasi, and Actinocamax alfridi.
Studying Insects: Entomology
Janet was especially interested in social insects like ants, wasps, and bees, which live in organized groups.
In 1894, he watched a hornet's nest from when it started until the last hornet worker died, which took five months! During this time, he discovered something called trophallaxis. This is when hornet larvae share food by exchanging liquid. He invented a special vertical artificial nest that became a very useful tool for scientists studying insects. This nest helped him understand how some insects, like silverfish, cleverly steal sugary liquid from ants.
He also studied the inside of ants' bodies in great detail. He showed how their bodies are organized in repeating segments, like a series of connected parts. He made an amazing discovery about young queen ants: after they tear off their wings, their flight muscles change into fat cells. These fat cells provide all the energy the queen needs to start her new colony without eating for many months!
Most of his research papers for the French Academy of Sciences were about social insects. He tried to connect how insects behave (ethology) with how their bodies work (physiology). A famous writer, Maurice Maeterlinck, praised Janet's work, saying his studies were "precise, clear, impeccable" and greatly enriched the study of ants and other sciences.
Understanding Atoms: Chemistry
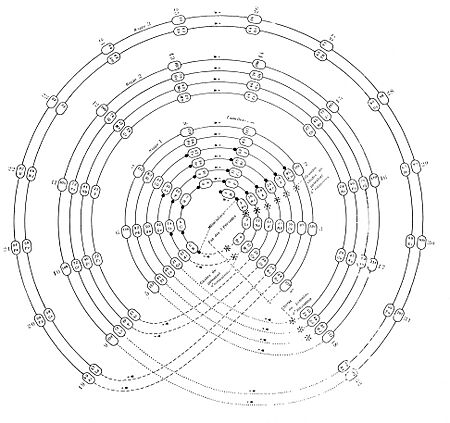
When Charles Janet was 78 years old, he started researching atoms, the tiny building blocks of everything. He was curious about their properties and how their centers (nuclei) were organized. To make sense of his ideas, he created his own way of arranging the chemical elements, a new periodic table. He believed that the physical and chemical properties of elements were closely linked to how they were arranged in his table.
Janet saw a perfect pattern in his table, which convinced him he had found the correct way to organize elements. In 1930, he even suggested checking his classification against the very new quantum theory, which describes how tiny particles behave. In doing so, he was the first to describe a rule for how electrons fill the different energy levels (subshells) around an atom. This rule was later rediscovered by others and is now known as the Madelung rule or the Klechkowski rule.
For a long time, Janet's classification was not well-known in France. However, thanks to his amazing spiral diagrams, it reappeared 40 years later among American chemists. Today, for about ten years, his "Left Step Table" has been seen as a good alternative to the famous Mendeleev's classification. An American historian named Eric Scerri has helped make Janet's work popular in science magazines, highlighting him as an important thinker in the history of chemistry.
Janet's Left-Step Periodic Table
Janet believed that the series of chemical elements is a continuous flow. He imagined this as a spiral shape drawn on the surfaces of four cylinders, one inside the other. By changing these shapes, he created several interesting designs, including his "left-step periodic table." In this table, elements like hydrogen and helium are placed above lithium and beryllium. He later realized that his arrangement matched perfectly with quantum theory and how electrons are structured in atoms.
Interestingly, he placed the actinides (a group of heavy elements) below the lanthanides (another group) twenty years before another scientist, Glenn Seaborg, did the same. He also continued his series up to element 120, predicting elements that had not yet been discovered.
Janet's table is different from the standard periodic table. It places the s-block elements (like hydrogen and helium) on the right side. This way, the different electron shells are arranged in a smooth order from left to right. This means there's no need to break the sequence or move the f-block elements (lanthanides and actinides) to a separate "footnote" section at the bottom, as is often done in the standard table. He thought that no elements heavier than number 120 would be found, so he didn't include a "g block" for even heavier elements.
His table also follows the Madelung rule, which explains the order in which electrons fill the different energy levels around an atom. The philosopher of chemistry Eric Scerri has strongly supported Janet's left-step periodic table, and it is now being discussed more and more as a potentially better or more fundamental way to organize the elements.
Template:Periodic table (left step)
Janet also imagined an "element zero" made of two neutrons. He thought this could be a link to a "mirror-image" table of elements with negative atomic numbers, almost like anti-matter. He also thought about "heavy hydrogen" (which we now call deuterium). Sadly, he passed away just before scientists discovered the neutron, the positron, and heavy hydrogen. His work was especially promoted by Edward G. Mazurs.
Charles Janet's Family
Charles Janet's brother, Armand Janet, was also an engineer and a scientist who studied insects. Armand became well-known for studying butterflies and moths (a lepidopterist) and was the president of the French Entomological Society in 1911.
Images for kids