White spruce facts for kids
Quick facts for kids White spruce |
|
|---|---|
 |
|
| Mature white spruce in Fairbanks, Alaska | |
| Conservation status | |
| Scientific classification |
|
| Kingdom: | Plantae |
| Clade: | Tracheophytes |
| Division: | Pinophyta |
| Class: | Pinopsida |
| Order: | Pinales |
| Family: | Pinaceae |
| Genus: | Picea |
| Species: |
P. glauca
|
| Binomial name | |
| Picea glauca (Moench) Voss
|
|
 |
|
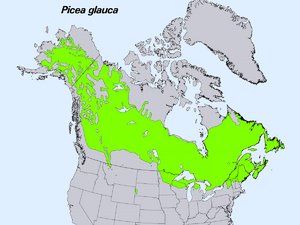
| Natural range | |
| Script error: The function "autoWithCaption" does not exist. | |
| Synonyms | |
|
List
|
|
Script error: No such module "Check for conflicting parameters".
| NCBI genome ID | 3330 |
|---|---|
| Ploidy | 2 |
| Genome size | 20 Gbp |
| Number of chromosomes | 12 |
| Year of completion | 2015 |
| Sequenced organelle | plastid and mitochondrion |
| Organelle size | 123 kbp and 5.93 Mbp |
| Year of completion | 2015 |
The white spruce (Picea glauca) is a type of spruce tree. It is native to the northern parts of North America. You can find it in the cool, northern boreal forests.
White spruce originally grew from central Alaska across Canada to Newfoundland. Now, it also grows naturally in some northern US states. These include Montana, Minnesota, Wisconsin, Michigan, Vermont, New Hampshire, and Maine. There's also a small group in the Black Hills of South Dakota and Wyoming. This tree has many other names, like Canadian spruce and Black Hills spruce.
Contents
What Does White Spruce Look Like?
The white spruce is a large evergreen tree. This means it keeps its needles all year. It usually grows about 15 to 30 meters (50 to 100 feet) tall. Some can even reach 40 meters (130 feet) tall! Its trunk can be up to 1 meter (3 feet) wide.
The tree's bark is thin and peels off in small, round pieces. Young trees have a narrow, cone-shaped top. Older trees have a more cylinder-shaped top. The branches have pale brown shoots. Its leaves are like needles, about 12 to 20 millimeters (0.5 to 0.8 inches) long. They are diamond-shaped when you look at them closely. They are blue-green on top, which is why it's called "glauca," meaning "bluish-green."
Cones and Seeds
The cones hang down from the branches. They are thin and cylinder-shaped, about 3 to 7 centimeters (1.2 to 2.8 inches) long. When closed, they are about 1.5 centimeters (0.6 inches) wide. They open up to about 2.5 centimeters (1 inch) wide. The cones have thin, flexible scales. They start green or reddish and turn pale brown after 4 to 8 months.
The seeds are small and black, about 2 to 3 millimeters (0.08 to 0.12 inches) long. Each seed has a thin, pale brown wing. This wing helps the seeds fly away in the wind. A single cone can hold between 32 and 130 good seeds.
Seeds are ready to spread in late summer or early fall. They usually travel by wind. Most seeds fall close to the parent tree. However, some can travel more than 300 meters (1,000 feet) away.
-
The bark is thin and scaly, flaking off in small circular plates
Roots and Trunk
The root system of a white spruce can change a lot. It adapts to different soil conditions. For example, if the soil is shallow, the roots might spread out like a plate.
White spruce trees can live for a very long time. Many live for 200 to 300 years. Some slow-growing trees in harsh climates can live even longer.
Where Does White Spruce Grow?

White spruce grows all across North America. In Canada, it covers almost all of the Boreal Forest region. It grows in every Canadian province and territory. It reaches as far north as 69°N latitude in the Mackenzie River delta. This is close to the Arctic Ocean.
In the United States, it grows in Maine, Vermont, New Hampshire, New York, Michigan, Wisconsin, Minnesota, and Alaska. In Alaska, it reaches the Bering Strait.
White spruce is one of the toughest conifers. It can handle very cold winter temperatures, sometimes as low as -56.5°C (-69.7°F). Its northern limit is often where the average July temperature is about 10°C (50°F). It needs a growing season of at least 60 days each year.

The southern edge of its range is more complex. Here, other trees like hardwoods compete with it. White spruce needs cold weather to grow well.
White Spruce in Other Countries
White spruce has been planted in other parts of the world, like England and parts of Europe. It was brought to England around 1700. It has also been planted in Tasmania and Sri Lanka. In Germany, it was used to help stabilize forests in windy areas. However, it hasn't always grown well in Britain because of damage from spring frosts.
How White Spruce Lives in Nature
White spruce is a "climax" tree in the boreal forests. This means it's one of the main trees that grow there over a long time. It usually grows in well-drained soils, especially near rivers. It can also grow in soils left by glaciers or lakes.
The ground under white spruce trees is often covered with feather mosses. In the far north, this moss and humus layer can be 25 to 46 centimeters (10 to 18 inches) deep.
White spruce can grow in soils with a pH from 4.7 to 7.0. It needs at least a moderate amount of nutrients in the soil to grow well. In the northern parts of its range, white spruce is a main species along with black spruce. Birch and aspen are often the first trees to grow after a disturbance like a fire.
Wildfires happen every 60 to 200 years in these forests. White spruce can grow in USDA Growing Zones 3-7. It does best in Zones 3-4, where it's colder. It doesn't like hot, humid weather.
The moss layer under white spruce trees can make the soil colder. This can even lead to permafrost (permanently frozen ground) in some areas.
Surviving the Cold
White spruce is very good at surviving cold temperatures. As long as the tree is ready for winter (in a state of "dormancy"), it can handle temperatures of -50°C (-58°F) or even colder. Its buds can survive temperatures down to -70°C (-94°F).
However, new shoots that grow in spring are very sensitive to late spring frosts. This can damage young trees.
How Forests Change Over Time
After a disturbance like a flood or fire, forests change over time. On new soil near rivers, you might first see shrubs. Then, trees like balsam poplar and white spruce start to grow.
On older floodplains, you often find a mix of white spruce and trembling aspen. Sometimes, after a fire, aspen and birch trees grow first. Then, white spruce comes in later. But if conditions are just right, white spruce can grow at the same time as the other trees.

The soil and light conditions affect how well white spruce grows. For example, birch trees can make it harder for spruce seedlings to grow.
Other Trees That Grow With White Spruce
White spruce often grows with other types of trees. In Alaska, it might grow with paper birch, trembling aspen, balsam poplar, and black spruce. In western Canada, you might also find it with subalpine fir, balsam fir, Douglas-fir, jack pine, and lodgepole pine.
The forest is usually dense, and the trees are well-shaped. The ground is often covered with mosses. There are usually not many other plants. However, some shrubs and herbs that grow there include alder, willows, cranberries, blueberries, and horsetail.
White spruce is a key tree in many forest types. It can grow in pure stands (only white spruce) or mixed stands (with other trees).
Pests and Diseases

Even though white spruce is strong, it can be attacked by insects and diseases. One major pest is the spruce budworm. This insect can cause a lot of damage. Other important insect pests include:
- Yellow-headed spruce sawfly
- European spruce sawfly
- Spruce bud moth
- Spruce beetle
Spruce beetles have destroyed huge areas of forests in Alaska. Many other insects, like different types of sawflies, loopers, aphids, and mites, can also harm white spruce trees.
How People Use White Spruce

White spruce is very important for its wood in Canada. The wood is used to make paper and for building. It's also a popular Christmas tree.
The wood is even sent to Japan. There, it's called "shin-kaya" and is used to make go boards. This is a substitute for a rare wood called kaya.
Wildlife also uses white spruce. Deer, rabbits, and grouse eat its needles in the winter.
The Black Hills Spruce (a type of white spruce) is used for bonsai, which is the art of growing miniature trees.
White spruce is the official provincial tree of Manitoba. It is also the state tree of South Dakota.
Different Types of White Spruce

Scientists have described several types of white spruce based on where they grow. These include:
- Picea glauca var. glauca: This is the typical white spruce, found from Newfoundland to eastern Alberta.
- Picea glauca var. densata (Black Hills white spruce): Found in the Black Hills of South Dakota.
- Picea glauca var. albertiana (Alberta white spruce): Found in the Rocky Mountains in Alberta, British Columbia, and Montana.
- Picea glauca var. porsildii (Alaska white spruce): Found in Alaska and Yukon.
The western types often have fuzzy shoots. White spruce can also mix with other spruce species. For example, it can hybridize with Sitka spruce where their ranges meet. This creates a hybrid known as Picea × lutzii.
Cultivated Varieties
Many special types of white spruce, called cultivars, have been grown for gardens. They come in different sizes, colors, and shapes. Some popular ones include:
- Picea glauca ‘Echiniformis’
- Picea glauca var. albertiana ‘Alberta Globe’
- Picea glauca var. albertiana ‘Conica’
'Conica' is a very small, or dwarf, conifer. It grows very slowly, only about 2 to 10 centimeters (0.8 to 4 inches) per year. Older 'Conica' trees sometimes start growing much faster and develop normal adult leaves. If you want to keep them small, you need to prune these faster-growing branches.
Images for kids
-
White spruce taiga along the Denali Highway in the Alaska Range
-
Mixed forest with balsam fir (Abies balsamea) at the southern edge of the white spruce range in Maine
-
White spruce growing in the riparian zone of the Sautauriski River, Québec
-
Old-growth Alaskan white spruce, Yukon
-
Choristoneura fumiferana, the eastern spruce budworm. Adult (silvery) and pupa (dark brown) on a (probably white) spruce.
-
Black Hills spruce grown as bonsai
-
Dwarf Alberta white spruce in Los Angeles County Arboretum and Botanic Garden






































