John Dalton facts for kids
Quick facts for kids
John Dalton
|
|
|---|---|

|
|
| Born | 6 September 1766 |
| Died | 27 July 1844 (aged 77) |
| Cause of death | Stroke |
| Nationality | British |
| Known for | Atomic theory, Law of Multiple Proportions, Dalton's Law of Partial Pressures, Daltonism |
| Awards | Royal Medal (1826) |
| Scientific career | |
| Notable students | James Prescott Joule |
| Influences | John Gough |
| Author abbrev. (botany) | Jn.Dalton |
| Signature | |
John Dalton (born September 6, 1766 – died July 27, 1844) was an English chemist, physicist, and weather scientist. He is famous for two main things. First, he brought the atomic theory into chemistry. This idea helps us understand what everything is made of. Second, he studied colour blindness. This condition is sometimes called Daltonism because of his work. Both John and his brother had colour blindness. He was the first to realize that this condition could be passed down in families.
Contents
Early Life and Education
John Dalton was born in Eaglesfield, England. His family belonged to a group called the Quakers. His father and a Quaker teacher named John Fletcher taught him when he was young. John's family was not rich, so he started working at age ten.
It is said that he began teaching at a local school when he was just 12 years old. By age 14, he was very good at Latin. When he was 15, John joined his older brother Jonathan. They ran a Quaker school together in Kendal.
John learned a lot about science from John Gough. Gough was a blind philosopher who was very talented in science and art. At 27, Dalton became a teacher of math and natural philosophy (which is like science) at a college in Manchester. He taught there for seven years. Then, he became a private tutor in math and science.
Dalton's Atomic Theory: Understanding Atoms
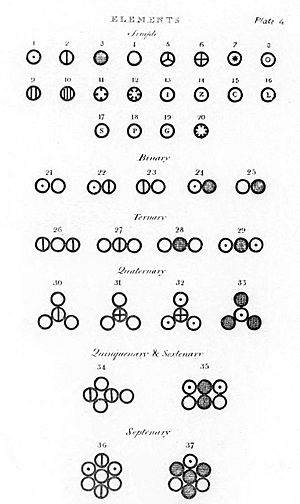
Dalton's Atomic Theory is a big idea in science about atoms. Atoms are the tiny building blocks of everything. Dalton created his theory to explain how elements combine in specific ways. The idea of atoms existed before Dalton. However, it was not widely accepted.
Dalton's theory was special because it was based on real observations. Before him, ideas about atoms were mostly based on philosophy. His work changed how scientists thought about matter.
Here are the main ideas of Dalton's atomic theory:
- Elements are made of very small particles called atoms.
- All atoms of one element are exactly alike in size, mass, and other features. Atoms of different elements are different.
- Atoms cannot be broken down, created, or destroyed.
- Atoms from different elements combine in simple, whole-number ways to form chemical compounds.
- During chemical reactions, atoms are joined, separated, or rearranged.
Dalton also used his own symbols to show how atoms connect. He drew these symbols in his book, New System of Chemical Philosophy. In this book, he listed 20 elements and 17 simple molecules.
John Dalton's Personal Life

John Dalton never got married. He had only a few close friends. As a Quaker, he lived a simple and quiet life. He did not seek fame or luxury.
For 26 years before he died, Dalton lived in a room in the home of Rev W. Johns. Johns was a botanist, someone who studies plants. Their home was on George Street in Manchester.
Dalton's daily life involved working in his laboratory and tutoring students. He would only take breaks for trips to the Lake District. He also sometimes visited London. In 1822, he went to Paris for a short time. There, he met many famous scientists. He also attended meetings of the British Science Association in different cities.
Health Challenges and Passing
Dalton had a small stroke in 1837. A second stroke in 1838 made it hard for him to speak. Even so, he could still do his experiments. In May 1844, he had another stroke. On July 26, 1844, he wrote down his last weather observation with a shaky hand. The next day, July 27, 1844, he fell from his bed in Manchester. His helper found him lifeless.
Dalton received a large public funeral. His body was displayed in Manchester Town Hall for four days. More than 40,000 people came to see his coffin. The funeral procession included people from the city's main groups. He was buried in Ardwick cemetery in Manchester. The cemetery is now a playing field.
John Dalton's Lasting Legacy


- Much of Dalton's written work was kept by a society in Manchester. Sadly, it was damaged during bombing in 1940.
- A statue of Dalton was put in Manchester Town Hall in 1877. He is thought to be the only scientist who had a statue made of him while he was still alive.
- A street in Manchester city centre is named after him: John Dalton Street.
- The John Dalton building at Manchester Metropolitan University is used by science and engineering students. A statue of Dalton stands outside it.
- A blue plaque marks the spot of his laboratory in Manchester.
- The University of Manchester has scholarships and a prize named after Dalton. A student residence hall is also called Dalton Hall.
- The Dalton Medal has been given out only twelve times by a Manchester society.
- A crater on the Moon was named after Dalton.
- "Daltonism" became a common word for colour blindness. In French, "daltonien" means "colour blind."
- The inorganic chemistry section of the UK's Royal Society of Chemistry is named after Dalton. Their journal for inorganic chemistry is also called Dalton Transactions.
- Many chemists use the term dalton (Da) to mean one atomic mass unit. This is 1/12 the weight of a neutral carbon-12 atom.
- Some Quaker schools have named buildings after Dalton. For example, a school house at Ackworth School is called Dalton.
- Dalton Township in Canada was named after him. Even though the township name changed, a new park was named Dalton Digby Wildlands Provincial Park in 2002.
Images for kids
See also
 In Spanish: John Dalton para niños
In Spanish: John Dalton para niños






