Snow facts for kids
Quick facts for kids Snow |
|
|---|---|

Norwegian train plowing through drifted snow
|
|
| Physical properties | |
| Density (ρ) | 0.1–0.8 g/cm3 |
| Mechanical properties | |
| Tensile strength (σt) | 1.5–3.5 kPa |
| Compressive strength (σc) | 3–7 MPa |
| Thermal properties | |
| Melting temperature (Tm) | 0 °C |
| Thermal conductivity (k) For densities 0.1 to 0.5 g/cm3 | 0.05–0.7 W/(K·m) |
| Electrical properties | |
| Dielectric constant (εr) For dry snow density 0.1 to 0.9 g/cm3 | 1–3.2 |
| The physical properties of snow vary considerably from event to event, sample to sample, and over time. | |

Snow is a type of ice. It forms when water in the air freezes. Snow can come in many different shapes and sizes.
Snow melts into liquid water at 0° Celsius (32° Fahrenheit). Sometimes, snow can turn directly into water vapor without melting first. This process is called sublimation. The opposite, when water vapor turns directly into snow, is called deposition.
Snow is popular for winter sports like skiing and sledding. People sometimes make artificial snow for these activities. It is also fun to build things out of snow, like snowmen or snow forts.
Snow can also be dangerous. It can make it hard to see and drive safely. When snow melts during the day and refreezes at night, it creates ice. This makes roads very slippery. Snowplows help clear roads. Also, sand or salt can be put on roads to help tires grip. Salt makes snow melt more easily because saltwater freezes at a lower temperature than fresh water.
A blizzard is a severe snowstorm. It has strong winds that blow snow around, making it very hard to see. Sometimes, blizzards can even have thundersnow, which means snow with lightning and thunder.
Contents
How Do Snowflakes Form?
Snow crystals form when tiny cloud droplets freeze. These droplets are very small, about 10 micrometers wide. They can stay liquid even when temperatures are below -18°C (0°F). For them to freeze, a few molecules in the droplet need to come together to form a tiny ice shape, called a "nucleus." The rest of the droplet then freezes around this nucleus.
This "homogeneous" freezing only happens at very cold temperatures, below -35°C (-31°F). In warmer clouds, a tiny dust particle or "ice nucleus" must be inside the droplet to help it freeze. Scientists are still learning which particles make the best ice nuclei. Clays, desert dust, and tiny living particles might work. People can also use artificial nuclei like silver iodide to make snow, a process called cloud seeding.
Once a droplet freezes, it grows in the air. This air is full of water vapor. Water molecules from the air stick to the ice crystal, making it bigger. Since there are many more water droplets than ice crystals, the ice crystals can grow very large, up to several millimeters. This process makes the water droplets evaporate, and the ice crystals grow at their expense.
These large crystals fall through the air. They can bump into each other and stick together, forming clusters called aggregates. These aggregates are what we call snowflakes. They are usually the type of ice that falls to the ground. The largest snowflake ever recorded was said to be 38 cm (15 inches) wide in Montana in 1887!
What Makes Snowflakes Unique?
Snowflakes are ice crystals with a special, symmetrical shape. Most snowflakes have six sides or points. This is because water molecules naturally arrange themselves into six-sided crystals when they freeze.
The shape of a snowflake depends on the temperature and humidity where it forms. For example, at about -2°C (28°F), snowflakes can sometimes form with three sides, looking like tiny triangles. Most snow particles you see are actually irregular, but perfect snowflakes are often shown in pictures because they look so beautiful.
- Between 0°C (32°F) and -3°C (27°F), snowflakes grow thin and flat.
- Between -3°C (27°F) and -8°C (18°F), they form long, thin shapes like needles or hollow columns.
- From -8°C (18°F) to -22°C (-8°F), they go back to being plate-like, often with many branches. This is where the most detailed snowflakes grow.
- Below -22°C (-8°F), they become column-like again.
If a snowflake starts growing as a column in one temperature zone and then falls into a warmer zone, it can grow new branches or plates at its ends. This creates "capped columns."
It is often said that no two snowflakes are exactly alike. However, in 1988, two matching snow crystals were found in Wisconsin. They were hollow hexagonal prisms, not the usual branched flakes.
Different Types of Snow

Snow can be described by how its flakes look, how fast it falls, and how it collects on the ground.
- Light snow means you can see more than 1 kilometer (0.6 miles).
- Moderate snow means visibility is between 0.5 km (0.3 mi) and 1 km (0.6 mi).
- Heavy snow means you can see less than 0.5 km (0.3 mi).
- A blizzard and snowstorm mean heavy snowfall with strong winds.
- Snow flurries are the lightest type of snow showers.
Sometimes, snow falls as small balls instead of flakes. This happens when snow melts and refreezes. These are called graupel, and types include sleet and snow pellets.
Once snow is on the ground, it can be:
- Powdery when it is light and fluffy.
- Granular when it starts to melt and refreeze.
- Ice when it packs down very densely after many melt-freeze cycles.
Powdery snow can drift with the wind, sometimes forming deep piles. If blown snow sticks to hillsides, it can form a snow slab, which is a risk for avalanches. Hoar frost can form on snow when winds are light and there is moisture in the air.
How Much Water is in Snow?
Snow stays on the ground until it melts or sublimates. The "water equivalent" of snow tells you how much water it would become if it melted. For example, 13 inches (33 cm) of fresh snow usually melts down to about 1 inch (2.5 cm) of water. This is because new snow is mostly air, with a density of about 8% of water.
Once snow is on the ground, it settles and becomes denser. Its density can reach about 30% of water. It gets even denser when it melts and refreezes, especially if temperatures rise above freezing or the sun shines on it. By late spring, snow can be as dense as 50% of water.
In places with mountains, melting snow in spring is a major source of water. This is very important for cities and farms downstream, especially in areas with dry summers. Scientists measure snow's water content to predict how much water will be available. If snow doesn't melt completely in the summer, it turns into firn, which is a type of old, dense snow. Firn can eventually become part of a glacier.
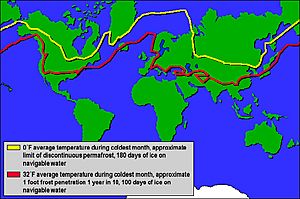
Where Does Snow Fall Most?

Fresh snow reflects over 90% of the sun's energy. This means it doesn't absorb much heat from the sun. Many rivers that start in mountains or cold regions get most of their water from melting snow. This can cause rivers to have very high flows at certain times of the year, sometimes leading to flooding.
The world record for the most snow in one season was at Mt. Baker Ski Area in Washington, USA. In 1998–1999, it received an amazing 2896 cm (1,140 inches) of snow!
How Snow Affects People
Heavy snowfall can cause big problems for cities and towns. It can slow down or stop air and ground travel. Even basic services like electricity and phone lines can fail. Roads become very difficult to use, and vehicles can get stuck.
Because of this, schools and workplaces sometimes close for a "snow day." In places that don't usually get much snow, even a small amount can cause a snow day because they are not ready for it.
In the past, people in snowy areas used to collect snow and store it in icehouses. They would cover it with straw or sawdust to keep it frozen. This ice could then be used in summer for cooling or medicine.
Snow and Farming
Snow can be good for farming. It acts like a warm blanket, protecting crops from very cold weather. Some farms rely on snow building up in winter to provide water when it melts in spring. If snow melts and refreezes on delicate crops like oranges, the ice can actually protect the fruit from even colder temperatures.
Snow and Your Eyes
Snow can be very bright. Snow blindness is a painful eye condition caused by too much ultraviolet (UV) light reflecting off snow or ice. Fresh snow reflects about 80% of UV light. This is a problem in very cold places and at high altitudes, because UV rays get stronger with elevation.
Snow also makes night skies much brighter. If there are clouds and snow is falling, the light from city lights reflects off the clouds and snow, making the night seem very bright. A similar effect happens on a full moon night with lots of snow.
Dangers of Snow
When heavy, wet snow falls on trees that still have their leaves in early autumn, it can cause a lot of damage. This kind of damage can be as bad as a hurricane.
An avalanche happens when too much snow builds up on a mountain and then suddenly slides downhill. This can be caused by a sudden change in temperature. Avalanches can be very destructive.
Large amounts of snow on buildings can also be dangerous. The weight of the snow can cause roofs to collapse.
Fun Things to Do in Snow

- Many winter sports like skiing, snowboarding, snowmobiling, and snowshoeing need snow. If there isn't enough natural snow, places use snow cannons to make it.
- Kids and adults can have fun riding on a sled or in a sleigh.
- Snow can help you explore new areas like forests or fields. Your footsteps leave a clear path, which can be helpful.
- Building snowmen is a classic snow activity. You make a human shape out of snow, often using a carrot for a nose and coal for eyes.
- Snow can be used to make snow cones, which are usually eaten in warmer weather.
- You can build snow forts for games like Capture the flag or for snowball fights.
- The world's biggest snowcastle, the SnowCastle of Kemi, is built every winter in Kemi, Finland.
- Michigan Technological University holds an annual Winter Carnival with a huge Snow Sculpture Contest.
- Some places even have snowball softball tournaments, using a bright orange softball and snow-filled bags for bases.
Images for kids
-
Frontal snowsquall moving toward Boston, Massachusetts
-
Cold northwesterly wind over Lake Superior and Lake Michigan creating lake-effect snowfall
-
Fresh snow beginning to metamorphose: The surface shows wind packing and sastrugi. In the foreground are hoar frost crystals, formed by refrozen water vapor emerging to the cold surface.
-
Snowmelt-induced flooding of the Red River of the North in 1997
-
Traffic stranded in a 2011 Chicago snowstorm.
-
Winter conditions on Ontario Highway 401 in Toronto due to a snowsquall.
-
Satellite view of the Indus River, showing snow in the Himalayas, which feeds it, and agricultural areas in Pakistan that draw on it for irrigation.
-
Arctic fox, a predator of smaller animals that live beneath the snow
-
Trucks plowing snow on a highway in Missouri
-
Bivouac of Napoleon's Grande Armée, during the winter retreat from Moscow
-
Finnish ski troops during the invasion of Finland by the Soviet Union
-
Army vehicles coping with snow during the Battle of the Bulge of World War II.
-
Navy SEALs training for winter warfare at Mammoth Mountain, California.
See also
 In Spanish: Nieve para niños
In Spanish: Nieve para niños