Light facts for kids
Light is a type of energy that we can see. It's a form of electromagnetic radiation that our eyes can detect. Light is just a small part of the whole electromagnetic spectrum. It comes from stars like the Sun, and many animals can see it too.
Light travels in tiny packets called photons. It acts like both a wave and a particle. Studying light is called optics, and it's a very important part of physics.
Light travels as a wave. Each wave has a specific wavelength or frequency. Our eyes see different wavelengths as different colors. For example, a rainbow shows all the colors of visible light. These colors are usually red, orange, yellow, green, blue, indigo, and violet.
There are other types of light we can't see. Wavelengths longer than red light are called infrared. Wavelengths shorter than violet light are called ultraviolet. Special cameras or instruments can detect these.
Contents
What is Light?
In empty space, light moves incredibly fast. This speed is called the speed of light, which is about 299,792,458 meters (or 186,282 miles) every second. This means it takes about 8 minutes for sunlight to reach Earth. When light travels through other materials, like glass, it slows down. For example, in glass, it moves about two-thirds as fast.
Light usually travels in a straight line. When something blocks its path, it creates shadows. Solid objects make darker shadows, while clearer objects make lighter ones. Transparent things, like clear glass, let light pass through easily, so they make very little or no shadow.
We see things because of light. Objects either give off their own light, like a lamp, or they reflect light from another source. For instance, everything else in a room reflects the light from a lamp.
Light and Color
Each color of light has a different wavelength. Shorter wavelengths mean the light has more energy. However, the speed of light doesn't change based on its energy.
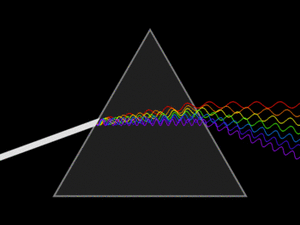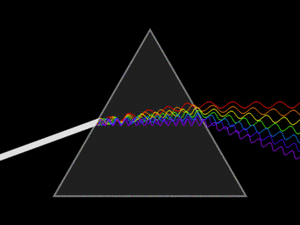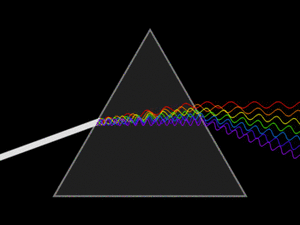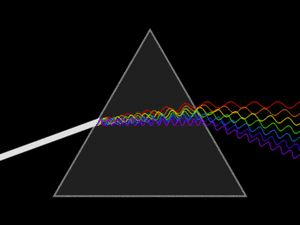
White light is actually a mix of many different colors of light. When white light passes through a prism, it splits into its separate colors, forming a spectrum. This spectrum includes all the wavelengths of light that our eyes can see. Red light has the longest wavelength, and violet (purple) light has the shortest.
Light with wavelengths shorter than violet is called ultraviolet light. Even shorter wavelengths include X-rays and Gamma rays. Light with wavelengths longer than red is called infrared light. Radio waves and microwaves (like those in a microwave oven) are also forms of electromagnetic radiation with even longer wavelengths. Our eyes can't see these, but special cameras can. All these different forms of light, visible and invisible, make up the electromagnetic spectrum.
When light bends in raindrops, a rainbow appears. The raindrops act like tiny prisms, bending the light and showing us the beautiful colors of the spectrum.
Our brains combine these three colors to create all the other colors we see.
Objects appear to have color because the tiny parts (molecules) that make them up absorb certain light waves. The light waves that are not absorbed bounce off the object. Our eyes see these reflected wavelengths, and our brain interprets them as a specific color.
How Fast Does Light Travel?
The speed of light in empty space is set at exactly 299,792,458 meters per second (about 186,282 miles per second). This exact number is used to define what a meter is. All types of electromagnetic radiation travel at this same speed in a vacuum.
Many physicists throughout history have tried to measure the speed of light.
- In 1676, a Danish physicist named Ole Rømer used a telescope to watch Jupiter and its moon, Io. He noticed that Io's orbit seemed to change slightly depending on Earth's position. He figured out that light took about 22 minutes to cross the diameter of Earth's orbit.
- In 1849, Hippolyte Fizeau in Europe used a spinning cog wheel and a mirror several kilometers away. He sent a light beam through a gap in the wheel, to the mirror, and back. By timing how fast the wheel spun for the light to return through the next gap, he calculated the speed of light.
- Léon Foucault used spinning mirrors in 1862 to get a more accurate measurement.
- Albert A. Michelson continued these experiments from 1877 until 1931. In 1926, he used improved spinning mirrors to measure the time light took to travel between Mount Wilson and Mount San Antonio in California. His very precise measurements gave a speed of 299,796,000 meters per second.
Light travels slower when it passes through transparent materials that contain ordinary matter. For example, light moves about three-quarters as fast in water as it does in empty space.
Scientists have even managed to slow light down a lot in special conditions. They passed light through a super-cold gas called a Bose–Einstein condensate. This doesn't mean the light completely stopped, but it was stored in the atoms and then re-emitted later.
Uses of Light on Earth
Sunlight is super important because it gives energy to green plants. Plants use this energy to make sugars (like starches) through a process called photosynthesis. These sugars then provide energy for almost all living things that eat plants or animals that eat plants.
Some animals can even make their own light! This is called bioluminescence. For example, fireflies use light to find mates, and some vampire squid use it to hide from predators.
Optics: The Study of Light

Optics is the study of light and how it interacts with different materials. Observing things like rainbows and the aurora borealis (northern lights) helps us understand more about light.
Refraction
Refraction is when light rays bend as they pass from one transparent material into another.
When a beam of light crosses from empty space into a material, or from one material to another, its wavelength changes. However, its frequency stays the same. If the light beam hits the boundary at an angle, this change in wavelength causes the light to change direction. This bending of light is what we call refraction.
The way lenses refract light is used to change how big images appear. Things like Magnifying glasses, spectacles, contact lenses, microscopes, and refracting telescopes all use this bending of light.
Where Does Light Come From?

There are many ways light is made:
- Heat: Hot objects give off light. The Sun is a great example; its surface is around 6,000 degrees Celsius and gives off a lot of visible light. Old-fashioned incandescent light bulbs also get very hot and glow, but they mostly produce infrared light (heat) and only a little visible light. Flames from a fire also glow because of hot particles.
* As an object gets hotter, the light it gives off changes color. It starts as a red glow, then turns white, and eventually blue-white as it gets extremely hot. You can see this when metal is heated in a forge.
- Atoms: Atoms can give off and absorb light at specific energies. This creates unique "emission lines" in the light spectrum of each atom. This is how light-emitting diodes (LEDs) and gas discharge lamps (like neon lamps and mercury-vapor lamps) work. Lasers also produce light this way.
- Moving Particles: Fast-moving charged particles, like electrons, can create visible light. This includes synchrotron radiation and bremsstrahlung. Also, if particles move faster than light can travel through a certain material, they can produce Cherenkov radiation, which is a blue glow.
- Chemical Reactions: Some chemical reactions produce light, a process called chemoluminescence. When this happens in living things, it's called bioluminescence, like the light from fireflies.
- Fluorescence and Phosphorescence: Some materials glow when brighter light shines on them (this is fluorescence). Others glow slowly after being exposed to light, even after the light source is removed (this is phosphorescence). This is used in old television sets and computer monitors.
Other ways light can be produced include:
- Bioluminescence (light from living things)
- Čerenkov radiation (light from fast particles in a medium)
- Electroluminescence (light from electricity)
- Scintillation (light flashes from radiation)
- Sonoluminescence (light from sound waves)
- Triboluminescence (light from friction)
If we talk about very high-energy light (like gamma rays), it can also be made by:
- Particles and antiparticles destroying each other
- Radioactive decay
Images for kids
-
A triangular prism dispersing a beam of white light. The longer wavelengths (red) and the shorter wavelengths (blue) are separated.
-
Pierre Gassendi was an early scientist who studied light.
-
Christiaan Huygens was another important scientist who studied light.
See also
 In Spanish: Luz para niños
In Spanish: Luz para niños