Periodic table facts for kids
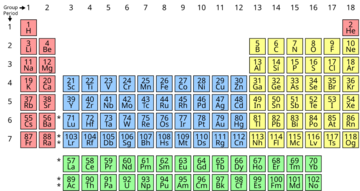
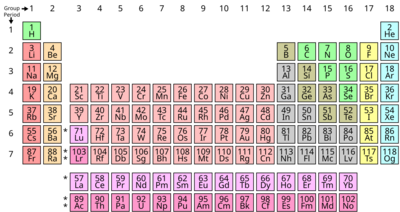
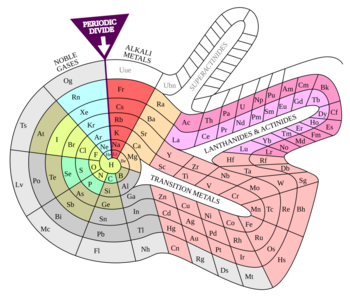
The periodic table is a special chart that organizes all the chemical elements we know. It arranges them into rows called "periods" and columns called "groups" based on their atomic numbers and how they behave. It's a fundamental tool used by scientists everywhere, from chemists to physicists, to understand how matter works.
This incredible table shows us a very important rule called the periodic law. This law tells us that when elements are lined up by their atomic numbers (which is like their unique ID number), their properties tend to repeat in a predictable way. It's like seeing a pattern in a song where certain notes come back again and again!
The table is divided into different sections, often called "blocks," and elements in the same column (or group) usually have similar chemical characteristics. This means they often react with other elements in similar ways. For example, some elements are shiny and good at conducting electricity (metals), while others are not (nonmetals). The periodic table helps us see these differences and similarities at a glance.
The periodic table keeps growing as science advances. As of February 2026, there are 118 known elements on the periodic table.
Contents
- The Building Blocks of Elements: Atoms and Their Parts
- How the Table is Organized: Periods and Groups
- History
- Understanding the Trends: Superpowers of Elements
- Special Groups and Families
- The Table Keeps Growing! Synthetic Elements and the Future
- Other Ways to See the Periodic Table
- Interesting facts about the Periodic Table
- Related Pages
- Images for kids
- See also
The Building Blocks of Elements: Atoms and Their Parts
To understand the periodic table, we first need to know a little about what makes up an element. Everything around us, from the air we breathe to the stars in the sky, is made of tiny particles called atoms.
- Atomic Number (Z): This is the most important number for an element! It tells you exactly how many protons are in the nucleus (the center) of an atom. Every element has a unique atomic number. For example, Hydrogen (H) always has 1 proton, so its atomic number is 1. Helium (He) always has 2 protons, so its atomic number is 2, and so on. This number is like the element's unique fingerprint!
- Protons: These are tiny, positively charged particles found in the atom's nucleus. The number of protons determines what element an atom is.
- Neutrons: These are also found in the nucleus, but they have no electrical charge (they're neutral). Neutrons add to an atom's weight but don't change its identity as an element.
- Electrons: These are tiny, negatively charged particles that whiz around the nucleus in special paths called "shells" or "orbitals." They are super important because they are the parts of the atom that interact with other atoms during chemical reactions!
- Chemical Symbol: Each element has a short, unique abbreviation, usually one or two letters. For example, H for Hydrogen, He for Helium, and Li for Lithium.
- Isotopes: Sometimes, atoms of the same element can have different numbers of neutrons. These variations are called isotopes. For example, carbon usually has 6 neutrons, but some carbon atoms have 7 or even 8 neutrons. They are all still carbon because they all have 6 protons! The periodic table groups all isotopes of an element together.
How the Table is Organized: Periods and Groups
The periodic table is arranged in a very clever way that helps scientists predict how elements will behave.
- Periods (Rows): These are the horizontal rows that go across the table. There are currently seven periods. A new period starts when a new "electron shell" begins to fill with electrons. Think of electron shells like layers of an onion around the nucleus. As you move from left to right across a period, the atomic number increases by one each time, meaning each element has one more proton and usually one more electron than the one before it.
- Groups (Columns): These are the vertical columns that go up and down the table. There are 18 groups. Elements in the same group tend to have similar chemical properties because they have the same number of electrons in their outermost shell (these are called valence electrons). These valence electrons are the ones involved in forming chemical bonds. For example, all elements in Group 1 (like Lithium, Sodium, and Potassium) are very reactive metals because they all have just one valence electron they are eager to share or give away.
| Group | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hydrogen & alkali metals |
Alkaline earth metals | Triels | Tetrels | Pnictogens | Chalcogens | Halogens | Noble gases |
||||||||||||
| Period |
|||||||||||||||||||
| 2 | |||||||||||||||||||
| 3 | |||||||||||||||||||
| 4 | |||||||||||||||||||
| 5 | |||||||||||||||||||
| 6 | |||||||||||||||||||
| 7 | |||||||||||||||||||
Primordial From decay Synthetic Border shows natural occurrence of the element
- Ca: 40.078 — Abridged value (uncertainty omitted here)
- Po: [209] — mass number of the most stable isotope
History
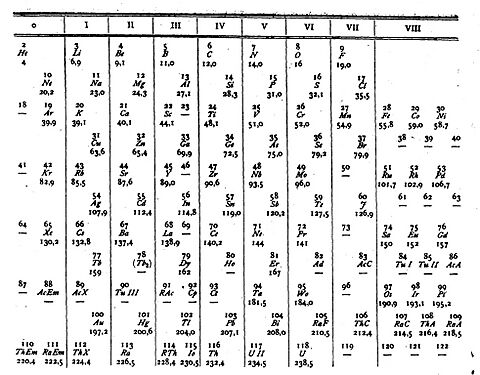
The idea of organizing elements isn't new! Scientists have been trying to make sense of them for a long time.
In the early 1800s, scientists like Johann Wolfgang Döbereiner] noticed that some elements could be grouped into "triads" (groups of three) with similar properties. Later, John Newlands proposed the "Law of Octaves," suggesting that properties repeated every eight elements, like musical notes. These were good starts, but they didn't work for all elements.
The real breakthrough came from a brilliant Russian chemist named Dmitri Mendeleev in 1869. He was the first to create a periodic table that was widely accepted. Mendeleev arranged the elements by their atomic weights, but here's the super cool part: he noticed gaps in his table! Instead of seeing these gaps as mistakes, he bravely predicted that these were places for elements that hadn't been discovered yet. He even predicted their properties, like their density and how they would react! In 1871, Mendeleev predicted the properties of three missing elements: "eka-boron," "eka-aluminium," and "eka-silicon." Just four years later, in 1875, French chemist Paul-Émile Lecoq de Boisbaudran discovered Gallium, which perfectly matched Mendeleev's "eka-aluminium"! In 1879, Swedish chemist Lars Fredrik Nilson discovered Scandium, which was Mendeleev's "eka-boron." And in 1886, German chemist Clemens Winkler found Germanium, which was "eka-silicon." Mendeleev's predictions were so accurate that they convinced the scientific world that his periodic law was a fundamental discovery!
Mendeleev used atomic weight, but later, scientists discovered something even better: the atomic number. In 1913, Dutch physicist Antonius van den Broek suggested that the number of positive charges in an atom's nucleus (the atomic number) was the key to its place on the table. English physicist Henry Moseley confirmed this experimentally in 1913-1914 using X-rays. He showed that elements should be ordered by their atomic number, not just their weight. This solved some puzzles where elements seemed out of place in Mendeleev's original table.
Danish physicist Niels Bohr, along with other scientists like Wolfgang Pauli, helped us understand that electrons orbit the nucleus in specific "shells" or energy levels. The way these electrons are arranged in shells explains why elements have their repeating properties. This understanding, based on the rules of quantum mechanics (the science of super tiny particles), truly explained why the periodic law works!
Understanding the Trends: Superpowers of Elements
The periodic table isn't just a list; it's a map that shows us how different properties of elements change in predictable ways. These are called periodic trends.
Atomic Radius (Size of Atoms)
- Across a Period (Left to Right): Atoms generally get smaller. Why? Because as you add more protons to the nucleus, the positive charge pulls the electrons in the same outer shell closer, making the atom shrink a bit.
- Down a Group (Top to Bottom): Atoms generally get larger. Why? Because as you go down, you're adding new electron shells, which are further away from the nucleus, making the atom bigger.
Ionization Energy
This is the energy needed to pull an electron away from an atom.
- Across a Period (Left to Right): It generally increases. Atoms on the right side hold onto their electrons more tightly because they have more protons pulling them in.
- Down a Group (Top to Bottom): It generally decreases. Electrons in larger atoms are further from the nucleus, so they are easier to pull away.
Electron Affinity
This is the energy released when an atom gains an electron.
- Across a Period (Left to Right): It generally increases. Atoms on the right side (especially the halogens) are very good at attracting extra electrons because they are close to having a full outer shell.
- Down a Group (Top to Bottom): It generally decreases. Larger atoms don't attract new electrons as strongly because the outer electrons are further from the nucleus.
When atoms share electrons to form a bond, electronegativity tells us how strongly an atom pulls those shared electrons towards itself.
- Across a Period (Left to Right): It generally increases. Elements like fluorine (the most electronegative reactive element) really want those electrons!
- Down a Group (Top to Bottom): It generally decreases. Larger atoms have less pull on shared electrons.
Metallicity (Metals, Nonmetals, and Metalloids)
This trend helps us classify elements based on their physical and chemical properties.
- Metals: These are usually found on the left and middle of the table. They are shiny, good conductors of heat and electricity, can be hammered into shapes (malleable), and stretched into wires (ductile). Examples: Iron, Gold, Copper.
- Nonmetals: These are usually found on the upper right side of the table. They are often dull, poor conductors, and brittle if solid. Examples: Oxygen, Carbon, Sulfur.
- Metalloids (or Semimetals): These elements sit on a diagonal line between metals and nonmetals. They have properties that are a mix of both! Examples: Silicon, Germanium. They are often used in electronics as semiconductors.
Special Groups and Families
The columns (groups) of the periodic table are often called "families" because the elements in them are related.
- 'Group 1: Alkali Metals (e.g., Lithium, Sodium, Potassium) – These are very reactive, soft, shiny metals that love to give away their single outermost electron.
- Group 2: Alkaline Earth Metals (e.g., Beryllium, Magnesium, Calcium) – Also reactive metals, but a bit less so than Group 1. They have two outermost electrons to share.
- Groups 3-12: Transition Metals (e.g., Iron, Copper, Gold) – These are the familiar metals we see every day. They are strong, shiny, and good conductors. Their electrons fill inner shells in a more complex way.
- Groups 13-16: Main-Group Elements (e.g., Carbon, Oxygen, Nitrogen) – This is a diverse group including metals, nonmetals, and metalloids.
- Group 17: Halogens (e.g., Fluorine, Chlorine, Iodine) – These are very reactive nonmetals that love to gain one electron to complete their outer shell.
- Group 18: Noble Gases (e.g., Helium, Neon, Argon) – These are the "lazy" elements! They have full outer shells, so they are very stable and usually don't react with other elements.
- Lanthanides and Actinides: These two special rows are usually placed at the bottom of the table to keep the main table from being too wide.
- Lanthanides (elements 57-71): These are often called "rare earth metals" and are very similar to each other. They are used in things like magnets and lasers.
- Actinides (elements 89-103): These are all radioactive, meaning their atoms are unstable and break down over time. Uranium and Plutonium, famous for nuclear energy, are actinides.
The Table Keeps Growing! Synthetic Elements and the Future
The periodic table isn't finished! Scientists are always working to discover or create new elements. The last elements to be officially named, Nihonium (113), Moscovium (115), Tennessine (117), and Oganesson (118), were given their names in 2016. This exciting event completed the entire seventh row of the periodic table!
Now, scientists are looking to the future, trying to create elements for an eighth period! As of 2026, scientists are actively trying to make element 119 at the Riken research institute in Japan (since 2018) and element 120 at the Lawrence Berkeley National Laboratory in the United States (since 2025). These super-heavy elements are incredibly difficult to make and are expected to be very unstable, lasting for only tiny fractions of a second.
Other Ways to See the Periodic Table
The standard periodic table is just one way to show the periodic law. Over the years, hundreds of different versions have been created! Some, like Charles Janet's left-step table, rearrange the blocks to highlight different patterns. Others are shaped like spirals, circles, or triangles.
These alternative tables help scientists emphasize different chemical or physical properties. While the standard table is the most common, scientists continue to explore if there's an "optimal" way to display all the elements.
Interesting facts about the Periodic Table
- Only elements up to atomic number 94 exist naturally.
- The remaining 24 elements (from Americium to Oganesson) are "synthetic," meaning they were created by scientists in laboratories!
- Most natural elements are stable, but some, like thorium and uranium, are radioactive.
- All 24 human-made elements are radioactive.
- Scientists predict there might be an "island of stability" in the periodic table, a region where some super-heavy elements could be more stable and last longer than others. Finding this island is a big goal for nuclear physicists.
Related Pages
Images for kids
See also
 In Spanish: Tabla periódica de los elementos para niños
In Spanish: Tabla periódica de los elementos para niños